Cobalt dichloride9/21/2023   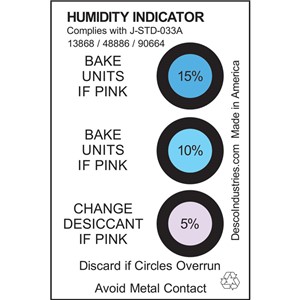
UsesĬobalt(II) chloride is used in humidity indicator in weather instruments. Solid lubricant, dye mordant, catalyst, barometers, UsesĪbsorbent for ammonia, gas masks, electroplating, sympathetic inks, hygrometers, manufacture of vitamin B It is also of use in manufacturing solid lubricants, as anĪdditive to fertilizers, as a chemical reagent in laboratories, and as an absorbent in gas masks,Įlectroplating, and the manufacture of vitamin B12. It is also used as a dye mordant (to fix the dye to Itself can cause damage to red blood cells. UsesĬobalt chloride (CoCl2) is used to manufacture vitamin B12, even though the compound Physical propertiesīlue leaflets turns pink in moist air hygroscopic the dihydrate is violet blue crystal the hexahydrate is pink monoclinic crystal density 3.36, 2.48 and 1.92 g/cm3 for anhydrous salt, dihydrate and hexahydrate, respectively anhydrous salt melts at 740☌ and vaporizes at 1,049☌ vapor pressure 60 torr at 801☌ the hexahydrate decomposes at 87☌ the anhydrous salt and the hydrates are all soluble in water, ethanol, acetone, and ether the solubility of hydrates in water is greater than the anhydrous salt. (1) Blue, (2) ruby-red crystals.Soluble in water, alcohol, and acetone. Pale blue leaflets, turns pink upon exposure to moist air. The anhydrous compound also may be obtained by passing chlorine over cobalt powder. The solution on concentration and cooling forms crystals of hexahydrate which on heating with SOCl2 dehydrates to anhydrous cobalt(II) chloride.Īlternatively, the hexahydrate may be converted to anhydrous CoCl2 by dehydration in a stream of hydrogen chloride and dried in vacuum at 100–150☌. The vapor-phase co-reductions with other metal halides by hydrogen results in finely divided intermetallics with applications as structural materials or compounds with useful thermoelectric, magnetic, and oxidation-resistance properties.Ĭobalt(II) chloride is prepared by the action of cobalt metal or its oxide, hydroxide, or carbonate with hydrochloric acid: It also is used to prepare several other cobalt salts and in the manufacture of synthetic vitamin B12. Humidity indicator as a temperature indicator in grinding as a foam stabilizer in beer in invisible ink for painting on glass in electroplating and a catalyst in Grignard reactions, promoting coupling with an organic halide. Water indicator, especially in desiccantsĬobalt dichloride is a strong allergen, with a LD 50 of 80 mg/kg (rats).Įye and hand protection should be worn while handling this compound.Ĭobalt(II) chloride should be stored in an airtight container or bag to counter its strong hygroscopic nature.Ĭobalt dichloride should be converted to an insoluble form before disposal.Cobalt(II) chloride has several applications.Cobalt chloride can be used for the synthesis of many complexes, such as Hexamminecobalt (III) chloride and Potassium cobaltinitrite.It can also be found in some types of colored silica gel, though the amount is small.Ĭobalt(II) chloride, along with other cobalt(II) compounds is classified as "Substance of very high concern" in the EU and its sale is regulated. If heptahydrate crystals are desired the solution may be dried in a desiccator.Ĭobalt(II) chloride can be purchased from chemical suppliers, both as a solid and as a solution. Usually, there will be a layer of unreacted cobalt carbonate/other contaminants that must be filtered out using either vacuum or gravity filtration in order to receive a pure product.īy heating this solution to dryness a light blue powder will be formed which is anhydrous cobalt chloride. Add a small amount of acid and then stir the mixture before adding another small portion, as the CO2 created can cause the mixture to overflow. The density for the three forms is 3.356 g/cm 3 (anhydrous), 2.477 g/cm 3 (dihydrate), 1.924 g/cm 3 (heptahydrate).Ĭobalt chloride heptahydrate can be synthesized by reacting two moles of hydrochloric acid with one mole of cobalt carbonate. CoCl 2 is soluble in water (52.9 g/100 mL at 20 ☌) methanol (38.5 g/100 mL), and slightly soluble in acetone, diethyl ether. The compound melts at 735 ☌ (anhydrous), 100 ☌ (dihydrate), 86 ☌ (hexahydrate). Cobalt(II) chloride is a salt that is blue when anhydrous and pink-purple when hydrated.
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
